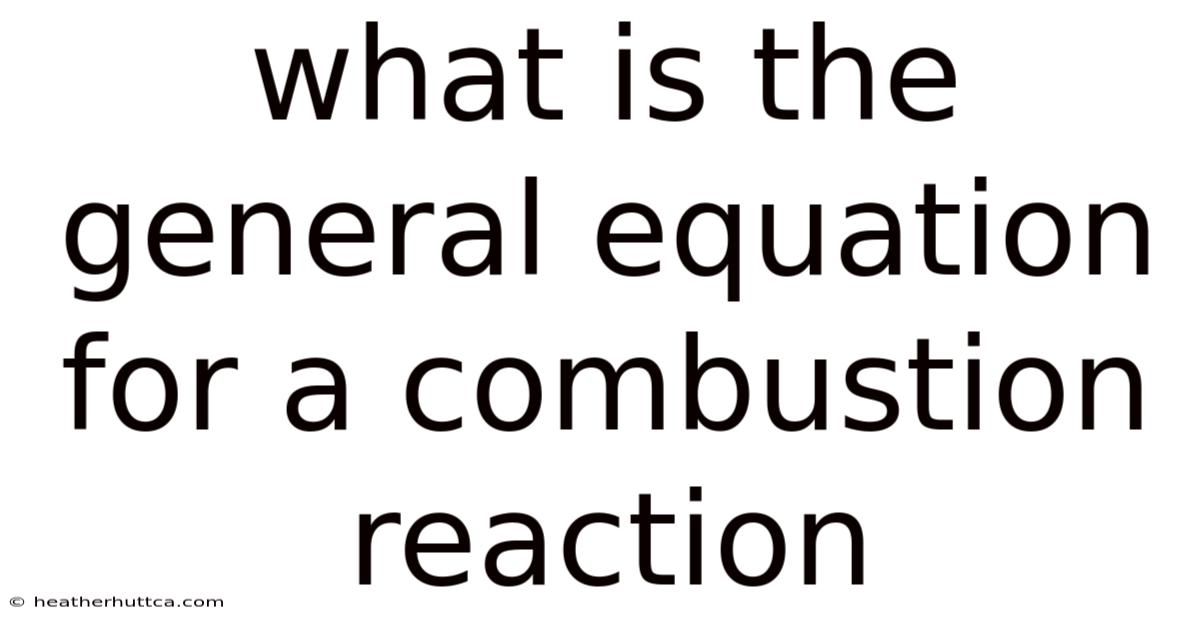The General Equation for a Combustion Reaction: A Complete Guide
Combustion reactions are the invisible engines of our modern world. From the flame that cooks your breakfast to the power that propels a jet across the sky, these chemical processes release immense energy by rapidly combining a fuel with oxygen. Now, at the heart of every combustion event—whether in a candle, a car engine, or a forest fire—lies a fundamental chemical pattern. Understanding the general equation for a combustion reaction is not just a textbook exercise; it is a key to comprehending energy production, environmental impact, and chemical safety. This equation provides a universal blueprint that allows scientists and engineers to predict products, calculate efficiency, and design cleaner technologies. This article will deconstruct that general formula, explore its variations, and illuminate the science behind one of humanity's most important chemical processes Most people skip this — try not to..
The Core Blueprint: The General Combustion Equation
The most common and fundamental type of combustion involves a hydrocarbon—a compound composed solely of hydrogen and carbon atoms, like methane (CH₄), propane (C₃H₈), or octane (C₈H₁₈)—reacting with oxygen (O₂) from the air. The general equation for a combustion reaction of a hydrocarbon is:
Fuel (Hydrocarbon) + Oxygen (O₂) → Carbon Dioxide (CO₂) + Water (H₂O)
In symbolic form, for a generic hydrocarbon with the formula CₓHᵧ: CₓHᵧ + O₂ → CO₂ + H₂O
This equation represents complete combustion, the ideal scenario where sufficient oxygen is present. On top of that, the process is highly exothermic, meaning it releases a large amount of heat and light. Now, the products are always carbon dioxide and water vapor. To make this equation chemically meaningful, it must be balanced according to the law of conservation of mass—the number of atoms of each element must be equal on both sides of the arrow. Balancing reveals the precise stoichiometric ratio of fuel to oxygen required.
To give you an idea, the balanced equation for methane (CH₄, where x=1, y=4) is: CH₄ + 2O₂ → CO₂ + 2H₂O Here, one molecule of methane reacts with two molecules of oxygen to produce one molecule of carbon dioxide and two molecules of water.
Complete vs. Incomplete Combustion: A Critical Distinction
The simplicity of the general equation masks a crucial reality: combustion is often incomplete. The type of combustion depends entirely on the oxygen supply Worth keeping that in mind. That's the whole idea..
Complete Combustion
Occurs when there is an excess of oxygen. The reaction proceeds to completion, converting all carbon to carbon dioxide (CO₂) and all hydrogen to water (H₂O). This process is:
- Cleaner: Produces fewer harmful byproducts.
- More Efficient: Releases the maximum possible energy from the fuel.
- Characterized by: A blue, hot flame (as seen in a well-adjusted Bunsen burner or gas stove).
Incomplete Combustion
Occurs when oxygen is limited. The reaction cannot fully oxidize all the fuel. Instead of CO₂, carbon-containing byproducts like carbon monoxide (CO)—a deadly, odorless gas—or even solid carbon (C) (soot) are formed. Water is still typically produced Simple, but easy to overlook. That alone is useful..
- Dirtier & More Dangerous: Produces toxic CO and particulate matter (soot).
- Less Efficient: Releases less total energy, as some fuel's chemical energy remains trapped in the partially oxidized products.
- Characterized by: A yellow, orange, or smoky flame (as seen in a candle flame or a poorly ventilated campfire).
The general equation for incomplete combustion of a hydrocarbon must account for these alternative products. Here's one way to look at it: if carbon monoxide is the primary carbon product: CₓHᵧ + O₂ → CO + H₂O (with insufficient O₂) If soot (carbon) is produced: CₓHᵧ + O₂ → C + H₂O
Combustion Beyond Hydrocarbons: Expanding the General Equation
While hydrocarbons are the most common fuels, the principle of combustion applies to other substances. The general equation adapts based on the elements in the fuel Easy to understand, harder to ignore..
- Combustion of Hydrogen (H₂): Hydrogen gas burns with oxygen to form only water. 2H₂ + O₂ → 2H₂O (This is the reaction that powered the Space Shuttle main engines).
- Combustion of Metals: Some metals, like magnesium (Mg), burn vigorously in oxygen. 2Mg + O₂ → 2MgO (Produces a brilliant white light, used in fireworks and flares).
- Combustion of Sulfur-Containing Fuels: Fossil fuels often contain sulfur. When they burn, sulfur dioxide (SO₂) is produced, a major contributor to acid rain. S + O₂ → SO₂
- Combustion of Alcohols: Methanol (CH₃OH) and ethanol (C₂H₅OH) are alcohols. Their combustion follows the hydrocarbon pattern but includes the oxygen atom already present in the fuel molecule. C₂H₅OH + 3O₂ → 2CO₂ + 3H₂O
The unifying theme is always oxidation (combination with oxygen) of a fuel, releasing energy. The specific products are determined by the fuel's elemental composition and the availability of oxygen.
The Stoichiometry Behind the Flame: Balancing the Equation
Balancing the general equation CₓHᵧ + O₂ → CO₂ + H₂O is a critical skill that reveals the exact oxygen-to-fuel ratio. Here is the systematic method:
- Balance Carbon (C): Place a coefficient of
xin front of CO₂ to balance thexcarbon atoms from the fuel. Consider this: cₓHᵧ + O₂ → xCO₂ + H₂O - Balance Hydrogen (H): There are
yhydrogen atoms in the fuel. To balance them, you needy/2molecules of H₂O (since each water molecule has 2 H atoms).
Continuing from thebalancing example:
-
Balance Oxygen (O): Count the oxygen atoms in the products (CO₂ and H₂O) and determine the required O₂ molecules. The coefficient for O₂ is found by dividing the total oxygen atoms needed by 2 (since each O₂ molecule provides 2 oxygen atoms) Which is the point..
-
Example (Methane, CH₄):
Products: CO₂ (1 O atom) + 2H₂O (4 O atoms) = 5 oxygen atoms total.
Required O₂: 5 oxygen atoms ÷ 2 atoms/O₂ = 2.5 O₂ molecules.
Balanced Equation: CH₄ + 2.5O₂ → CO₂ + 2H₂O -
Example (Ethane, C₂H₆):
Products: 2CO₂ (4 O atoms) + 3H₂O (6 O atoms) = 10 oxygen atoms total.
Required O₂: 10 ÷ 2 = 5 O₂ molecules.
Balanced Equation: C₂H₆ + 5O₂ → 2CO₂ + 3H₂O
-
This systematic approach ensures the law of conservation of mass is upheld, providing the precise oxygen-to-fuel ratio necessary for complete combustion to CO₂ and H₂O. This ratio is fundamental to designing efficient engines, furnaces, and power plants, maximizing energy output while minimizing fuel waste.
The Significance of Combustion Chemistry
Understanding the principles and equations of combustion – from the dangers of incomplete combustion to the precise stoichiometry of complete combustion – is crucial. It underpins:
- Energy Production: Optimizing fuel efficiency and maximizing energy yield from coal, oil, natural gas, biofuels, and hydrogen.
- Environmental Protection: Minimizing harmful emissions (CO, soot, SO₂, NOx) by ensuring complete combustion and implementing effective pollution control technologies.
- Material Science & Safety: Predicting and controlling combustion behavior in industrial processes, welding, rocketry, and fire safety.
- Sustainability: Guiding the development and utilization of cleaner fuels and more efficient combustion technologies to reduce our environmental footprint.
The general equation Cₓ
The generalequation CₓHᵧ + O₂ → CO₂ + H₂O exemplifies how stoichiometric precision underpins the practical applications of combustion chemistry. By ensuring every atom is accounted for, this balance allows engineers and scientists to predict reaction outcomes, optimize fuel usage, and mitigate risks associated with combustion processes. This foundational knowledge is not merely academic; it drives innovation in cleaner energy solutions, such as hydrogen fuel cells or advanced biofuels, where exact oxygen-to-fuel ratios minimize waste and maximize efficiency.
Conclusion
Combustion chemistry, though rooted in fundamental principles, has far-reaching implications that touch nearly every aspect of modern life. From the humble candle flame to the complex engines powering our transportation and industries, the balance of oxygen and fuel dictates efficiency, safety, and environmental impact. As global demand for sustainable energy grows, the lessons from stoichiometry and combustion reactions will remain vital. They challenge us to innovate beyond traditional fuels, embrace novel technologies, and harness chemistry as a tool for both progress and planetary stewardship. Understanding combustion is not just about mastering equations—it’s about unlocking the potential to create a safer, cleaner, and more sustainable future Practical, not theoretical..