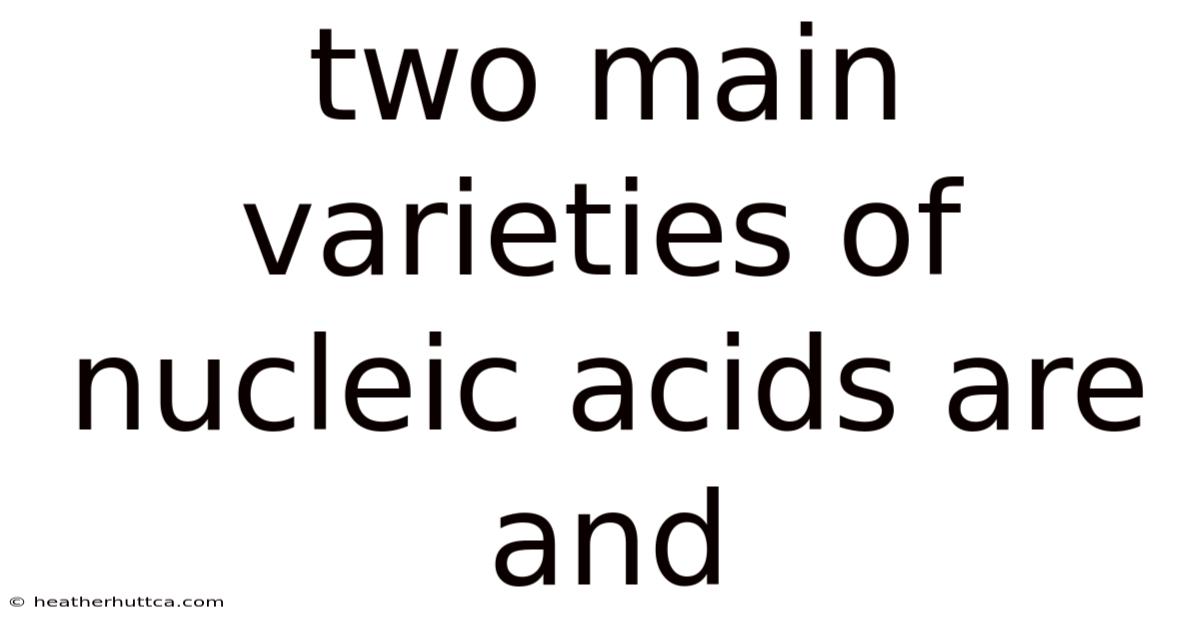Thetwo main varieties of nucleic acids are DNA (deoxyribonucleic acid) and RNA (ribonucleic acid), the fundamental polymers that store, transmit, and regulate genetic information in all known life forms. This article explores their structural distinctions, biochemical roles, and the molecular mechanisms that make each uniquely suited to its biological tasks, providing a clear, SEO‑optimized guide for students, educators, and anyone curious about the chemistry of heredity Worth keeping that in mind..
1. Structural Foundations
1.1 DNA – The Blueprint of Life
DNA consists of long chains of nucleotides linked by phosphodiester bonds. Each nucleotide contains a deoxyribose sugar, a phosphate group, and one of four nitrogenous bases: adenine (A), thymine (T), guanine (C), or cytosine (G). The double‑helix model, proposed by Watson and Crick, reveals two antiparallel strands that coil around each other, stabilized by hydrogen bonds between complementary bases (A‑T and G‑C). This helical structure protects the genetic code while allowing efficient replication Worth keeping that in mind..
1.2 RNA – The Versatile Messenger
RNA also polymerizes from nucleotides, but its backbone incorporates a ribose sugar and the base uracil (U) in place of thymine. RNA typically exists as a single‑stranded molecule that can fold into complex three‑dimensional shapes, forming hairpins, loops, and bulges. The three most common RNA types—messenger RNA (mRNA), transfer RNA (tRNA), and ribosomal RNA (rRNA)—exemplify diverse functional adaptations of the same basic polymer.
2. Functional Specializations
2.1 DNA’s Role in Information Storage
DNA serves as the primary repository of hereditary instructions. Its stable double‑helix conformation and ability to repair damaged strands make it ideal for long‑term archival of genetic data. During cell division, DNA undergoes replication, a semi‑conservative process that copies each strand to preserve the original genetic message with high fidelity.
2.2 RNA’s Multifaceted Functions
RNA translates the static genetic code into functional products:
- mRNA carries the coded instructions from the nucleus to ribosomes, where proteins are synthesized.
- tRNA acts as an adaptor, delivering specific amino acids to the ribosome in accordance with the mRNA codon sequence.
- rRNA forms the core of ribosomal subunits, catalyzing peptide bond formation and ensuring accurate translation.
Beyond protein synthesis, RNA participates in gene regulation (e.g., microRNA, siRNA), catalysis (ribozymes), and immune defense (RNA interference pathways) Less friction, more output..
3. Comparative Overview
| Feature | DNA | RNA |
|---|---|---|
| Sugar | Deoxyribose | Ribose |
| Strand Number | Typically double‑stranded | Usually single‑stranded |
| Base Composition | A, T, G, C | A, U, G, C |
| Structural Stability | High (due to lack of 2′‑OH) | Lower (2′‑OH makes it more reactive) |
| Primary Role | Long‑term genetic storage | Transient information transfer and catalysis |
| Replication | Occurs in the nucleus (or cytoplasm in prokaryotes) | Transcribed from DNA; may also replicate in some viruses |
These contrasts underscore why DNA is suited for permanent genetic archiving, while RNA excels in dynamic processes that require rapid synthesis and degradation.
4. Molecular Mechanisms Behind the Differences
4.1 Chemical Stability
The absence of a 2′‑hydroxyl group in deoxyribose renders DNA chemically inert, protecting it from hydrolysis. Conversely, the ribose sugar in RNA is more susceptible to alkaline hydrolysis, which aligns with RNA’s transient nature Practical, not theoretical..
4.2 Hydrogen‑Bonding Patterns
DNA’s complementary base pairing (A‑T via two hydrogen bonds, G‑C via three) creates a highly stable duplex. RNA’s pairing with uracil (A‑U) involves only two hydrogen bonds, contributing to its generally lower melting temperature and greater flexibility Easy to understand, harder to ignore..
4.3 Enzymatic Interactions
DNA polymerases replicate DNA with proofreading activity, ensuring high fidelity. RNA polymerases lack strong proofreading, resulting in a higher mutation rate that fuels viral evolution and rapid adaptation.
5. Biological Implications
5.1 Evolutionary Advantage
The stability of DNA enables organisms to maintain complex genomes across generations, supporting multicellularity and detailed developmental pathways. RNA’s relative instability allows quick responses to environmental cues, such as stress‑induced gene expression changes And it works..
5.2 Medical Relevance
Understanding the distinct properties of DNA and RNA underpins many therapeutic strategies:
- Gene therapy delivers corrected DNA sequences to fix genetic defects.
- RNA interference (RNAi) therapeutics silence disease‑associated genes.
- mRNA vaccines (e.g., COVID‑19 vaccines) exploit synthetic mRNA to trigger immune responses without altering the host genome.
6. Frequently Asked Questions
**Q1: Can
The interplay between catalytic precision and regulated response shapes biological evolution, highlighting RNA's dual role as both tool and shield.
Conclusion
Understanding these dualities enriches our grasp of life's nuanced mechanisms, bridging molecular functions with functional outcomes. Such insights guide advancements in biotechnology and medicine, underscoring the profound interconnectedness of nature's design.
Q1: Can RNA act as a permanent genetic material?
In most cellular life forms, RNA is not the primary repository of genetic information. Still, a few viruses—most notably the retroviruses (e.g., HIV) and many RNA viruses (influenza, SARS‑CoV‑2)—store their entire genome as RNA. In these cases, the virus must either reverse‑transcribe its RNA into DNA (retroviruses) or rely on high‑fidelity RNA‑dependent RNA polymerases (RNA viruses) to maintain the genome across replication cycles. The inherent instability of RNA limits the length and complexity of such genomes, which is why the largest known viral RNA genomes (coronaviruses) are still under 30 kb, far smaller than the megabase‑scale chromosomes of eukaryotes Most people skip this — try not to..
Q2: Why does RNA have a higher mutation rate than DNA?
Two main factors drive this difference:
- Lack of proofreading – RNA polymerases generally lack the 3′→5′ exonuclease activity that DNA polymerases use to excise misincorporated nucleotides.
- Chemical vulnerability – The 2′‑OH group makes RNA more prone to spontaneous hydrolysis and to damage from reactive oxygen species.
These features give RNA a “fast‑forward” evolutionary mode, which is advantageous for viruses that must outpace host immune defenses but disadvantageous for organisms that need long‑term genomic integrity Nothing fancy..
Q3: How do cells protect their RNA from degradation?
Cells employ several strategies:
- RNA‑binding proteins (RBPs) cloak transcripts, shielding them from ribonucleases.
- 5′ capping and 3′ polyadenylation of mRNA add protective structures that block exonuclease access and promote translation.
- Secondary structures (e.g., hairpins in tRNA and rRNA) confer intrinsic stability.
- Compartmentalization—for instance, the nucleolus concentrates rRNA synthesis and processing away from cytoplasmic nucleases.
Q4: Are there therapeutic approaches that exploit the instability of RNA?
Yes. Antisense oligonucleotides (ASOs) and small interfering RNAs (siRNAs) are designed to bind specific mRNA targets, recruiting RNase H or the RNA‑induced silencing complex (RISC) to degrade the transcript. Because RNA turnover is rapid, these interventions can achieve swift knock‑down of disease‑causing proteins without permanent genome alteration.
7. Emerging Frontiers: Blurring the DNA–RNA Divide
7.1 Reverse Transcription and Retrotransposition
Retrotransposons—mobile genetic elements that comprise nearly half of the human genome—transpose via an RNA intermediate that is reverse‑transcribed back into DNA. This process demonstrates how RNA can temporarily serve as a genetic carrier before re‑entering the DNA repository, illustrating a natural “copy‑and‑paste” mechanism that fuels genomic innovation (and disease when dysregulated).
7.2 RNA‑Based Epigenetics
Beyond the classic DNA methylation and histone modifications, cells employ RNA modifications (the epitranscriptome) such as N⁶‑methyladenosine (m⁶A) to fine‑tune transcript stability, splicing, and translation. These reversible marks act analogously to DNA epigenetic marks, providing a rapid, reversible layer of gene regulation that can respond to metabolic cues, stress, or developmental signals.
7.3 Synthetic Biology and Xeno‑Nucleic Acids (XNAs)
Researchers have engineered nucleic acids with altered backbones (e.g., HNA, TNA) that combine DNA’s stability with RNA’s functional versatility. XNAs can store information like DNA while folding into catalytic structures reminiscent of ribozymes, opening possibilities for orthogonal genetic systems that are resistant to natural nucleases—an attractive platform for biocontainment and novel therapeutics.
8. Practical Takeaways for Researchers and Clinicians
| Area | Key Insight | Application |
|---|---|---|
| Molecular Diagnostics | RNA’s rapid turnover mirrors real‑time cellular states. On top of that, g. | Design modular mRNA platforms for emerging pathogens; adapt codon usage and UTRs for optimal expression. |
| Gene Editing | DNA editing provides permanent correction; RNA editing offers reversible modulation. Day to day, | |
| Vaccine Development | mRNA can be produced quickly and induces strong immune responses without integration risk. g. | Develop nucleoside analogs (e. |
| Antiviral Strategies | Targeting viral RNA replication circumvents the need for host genome alteration. On the flip side, cRISPR‑Cas13 or ADAR‑based editors for transient phenotypic rescue. , remdesivir) that stall viral RNA‑dependent RNA polymerases. |
Conclusion
DNA and RNA, though built from the same nucleotide alphabet, have diverged dramatically in structure, stability, and biological purpose. DNA’s solid, double‑helical architecture makes it the ideal long‑term vault for genetic information, while RNA’s single‑stranded flexibility, catalytic potential, and rapid turnover empower cells to sense, respond, and adapt on the fly. The nuanced interplay between these two polymers underlies everything from the faithful inheritance of complex multicellular organisms to the swift evolution of RNA viruses Still holds up..
It sounds simple, but the gap is usually here.
By dissecting the chemical underpinnings—sugar chemistry, hydrogen‑bonding patterns, and enzymatic fidelity—we gain a mechanistic appreciation for why nature has assigned each nucleic acid its distinct role. Worth adding, modern science is increasingly learning to repurpose these traits: leveraging DNA’s permanence for gene therapy, exploiting RNA’s transience for vaccines and gene silencing, and even engineering hybrid systems that blur the traditional boundaries.
In sum, the dichotomy of DNA versus RNA is not a rigid opposition but a complementary partnership that fuels life’s continuity and its capacity for change. Recognizing and harnessing this partnership continues to drive breakthroughs in medicine, biotechnology, and our fundamental understanding of biology.