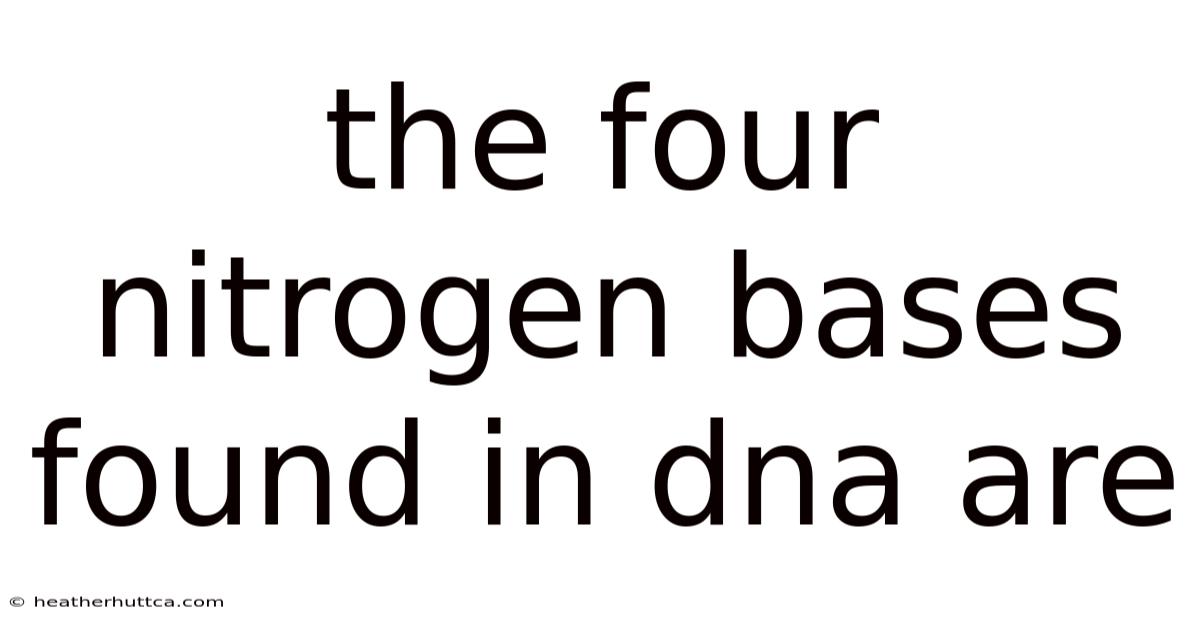The four nitrogen bases found in DNAare adenine, thymine, cytosine, and guanine, the molecular building blocks that encode genetic information and dictate the hereditary traits of all living organisms. Understanding these bases is essential for grasping how DNA stores, replicates, and transmits the instructions that shape life, from the simplest bacteria to complex humans. This article explores the chemical nature of each base, their complementary pairing rules, the role they play in the double‑helix structure, and the broader biological implications of their interactions.
Introduction In the realm of molecular biology, the term nitrogen base refers to the organic molecules that protrude from the sugar‑phosphate backbone of DNA. These bases are not merely passive components; they are active participants in the storage and expression of genetic code. The four nitrogen bases found in DNA are adenine (A), thymine (T), cytosine (C), and guanine (G). Their unique structures enable precise pairing, ensuring accurate replication and transcription. By examining each base individually and collectively, we can appreciate how they underpin the stability, fidelity, and functionality of the genetic material.
The Four Nitrogen Bases: Chemical Identity and Properties
Adenine
Adenine is a purine composed of a double‑ring structure that contains nitrogen atoms at positions 1, 3, 7, and 9. That's why in DNA, adenine extends outward from the backbone and forms hydrogen bonds with thymine, creating a stable A‑T pair. Day to day, its molecular formula is C₅H₅N₅, and it appears as a pale yellow crystalline solid. The presence of adenine in codons determines which amino acid will be incorporated during protein synthesis.
Thymine
Thymine is a pyrimidine with the molecular formula C₅H₆N₂O₂. Because of that, unlike adenine, thymine possesses a methyl group at the fifth carbon, which enhances its hydrophobic character and contributes to the overall stability of the DNA double helix. Thymine pairs with adenine through two hydrogen bonds, a relationship that is crucial for maintaining the regular spacing of the helix Small thing, real impact. Less friction, more output..
Cytosine
Cytosine is another pyrimidine, with the formula C₄H₅N₃O. It lacks the methyl group present in thymine, making it slightly more polar and prone to deamination, a process that can lead to mutations if not repaired. Cytosine forms three hydrogen bonds with guanine, resulting in a stronger interaction than the A‑T pair and contributing to the overall stability of the DNA strand Less friction, more output..
Guanine
Guanine is the final purine base, with the molecular formula C₅H₅N₅O. So its structure includes an amino group attached to the second carbon of the imidazole ring, which participates in hydrogen bonding with cytosine. The G‑C pair is characterized by three hydrogen bonds, giving it a higher binding energy than the A‑T pair and influencing the melting temperature of DNA segments.
Complementary Base Pairing: The Rules of the Genetic Code
The four nitrogen bases found in DNA are not random; they follow strict pairing rules that ensure accurate replication. These rules, discovered by Watson and Crick, are:
- Adenine pairs with Thymine (A‑T) – two hydrogen bonds hold them together.
- Cytosine pairs with Guanine (C‑G) – three hydrogen bonds stabilize this pair.
This complementary pairing is often visualized as a ladder where the rungs consist of these base pairs. The specificity of these interactions prevents mismatches that could disrupt the genetic code. During DNA replication, each strand serves as a template for the synthesis of a new complementary strand, a process that relies on DNA polymerases recognizing and incorporating the correct bases.
Scientific Explanation of Base Pairing and Stability
The stability of DNA is directly linked to the hydrogen‑bonding patterns between bases. The A‑T pair, with its two hydrogen bonds, is easier to break than the C‑G pair, which has three. Which means consequently, regions rich in G‑C content have higher melting temperatures, meaning they require more heat to separate the strands. This property influences gene regulation, as certain promoters are located in GC‑rich areas that are more stable and less prone to spontaneous unwinding.
On top of that, the hydrophobic effect plays a role: the stacked bases are held together by van der Waals forces, which are enhanced when the bases are planar and aromatic. The π‑stacking interactions between adjacent base pairs contribute significantly to the overall rigidity of the double helix, protecting the genetic information from mechanical stress Simple, but easy to overlook..
Biological Significance of the Four Nitrogen Bases
The four nitrogen bases found in DNA are integral to numerous cellular processes:
- Gene Expression: During transcription, the DNA template is read by RNA polymerase, which synthesizes messenger RNA (mRNA) using complementary bases. The sequence of bases in mRNA determines the amino‑acid sequence of proteins through the genetic code.
- DNA Repair Mechanisms: Cells possess enzymes that recognize mismatched bases and excise damaged segments, preventing mutations. The specificity of base pairing allows repair systems to distinguish between correct and incorrect nucleotides.
- Mutagenesis: Environmental factors such as radiation or chemicals can cause deamination of cytosine to uracil, leading to C‑G to T‑A transitions if unrepaired. Understanding these processes highlights the importance of maintaining base integrity.
- Evolutionary Diversity: Variations in base composition across species affect gene regulation and protein function, driving evolutionary adaptation. Take this case: high GC content in thermophilic organisms contributes to DNA stability at elevated temperatures.
Frequently Asked Questions (FAQ)
What distinguishes a purine from a pyrimidine?
Purines (adenine and guanine) consist of a double‑ring structure, whereas pyrimidines (cytosine, thymine, and uracil) have a single ring. This structural difference influences their hydrogen‑bonding capabilities and overall size within the DNA helix It's one of those things that adds up..
Why does thymine appear only in DNA and not in RNA?
Thymine differs from uracil (the RNA counterpart) by the presence of a methyl group at the fifth carbon. This methyl group enhances the stability of the DNA double helix and reduces the likelihood of deamination errors, making thymine more suitable for the more stable, long‑term storage of genetic information in DNA.
How do mutations arise from errors in base pairing?
If a DNA polymerase incorporates the wrong base opposite a template strand, the resulting mismatch may persist if not corrected by proofreading or mismatch‑repair mechanisms. Over time, such errors can become permanent mutations, potentially
altering protein function or disrupting regulatory elements. The cumulative effect of these changes can influence an organism's fitness, either negatively through disease or positively through advantageous adaptations And that's really what it comes down to..
The Interplay Between Base Structure and Genetic Fidelity
The specific geometry of each base ensures precise alignment within the helix, minimizing steric clashes and optimizing hydrogen‑bond formation. This structural precision is complemented by the hydrophobic stacking forces that shield the bases from the aqueous environment, further stabilizing the double helix. Because of this, the molecule achieves a balance between flexibility for replication and rigidity for information preservation No workaround needed..
At its core, the bit that actually matters in practice Not complicated — just consistent..
Conclusion
The four nitrogenous bases of DNA—adenine, guanine, cytosine, and thymine—form the foundational code of life through their precise pairing and structural organization. Their chemical properties enable not only the faithful storage of genetic information but also the dynamic processes of replication, repair, and expression. By understanding the involved roles of these molecules, we gain insight into the fundamental mechanisms that govern inheritance, evolution, and the very continuity of life.