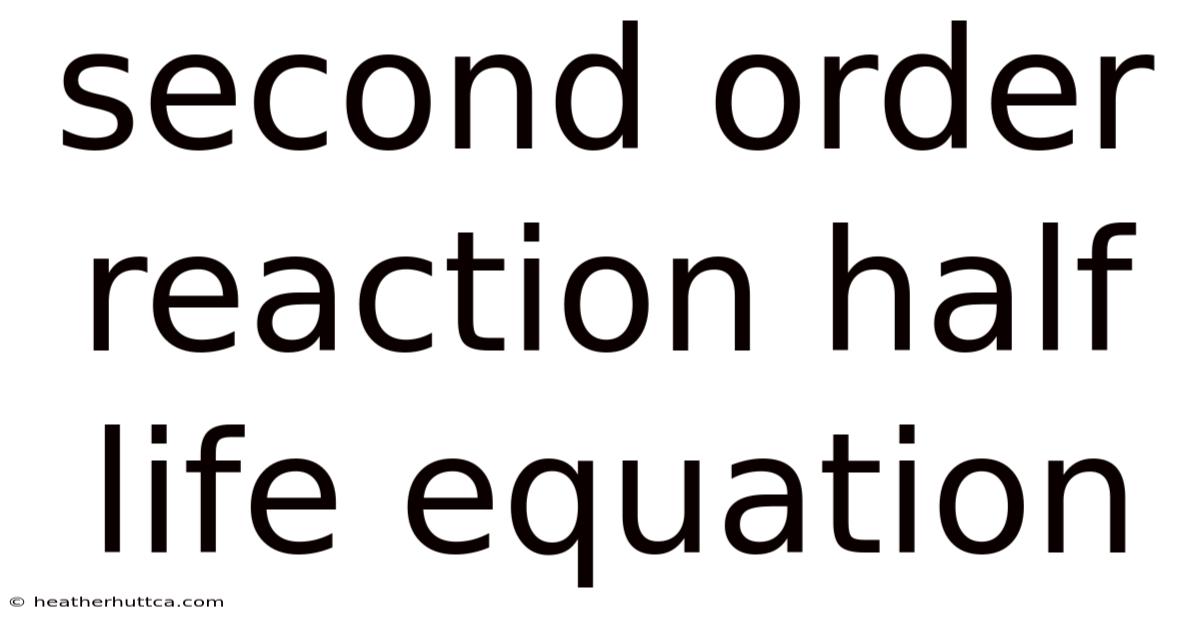The Second Order Reaction Half Life Equation: Understanding Concentration-Dependent Kinetics
The second order reaction half life equation is a cornerstone of chemical kinetics, offering insights into how reaction rates evolve over time when the rate depends on the square of a single reactant’s concentration or the product of two reactants’ concentrations. This equation is important for predicting how long it takes for a reactant’s concentration to halve, a concept known as half-life. Unlike first-order reactions, where half-life remains constant regardless of initial conditions, second-order reactions exhibit a dynamic relationship between concentration and time. This dependency makes the second order reaction half life equation indispensable for chemists, engineers, and researchers working with processes where reactant concentrations significantly influence reaction speed.
At its core, a second-order reaction is defined by its rate law, which states that the reaction rate is proportional to the concentration of one reactant raised to the second power or the product of two reactants’ concentrations. To give you an idea, a reaction involving a single reactant A might follow the rate law rate = k[A]², while a bimolecular reaction between A and B could follow rate = k[A][B]. The second order reaction half life equation emerges from integrating these rate laws, revealing how initial concentrations dictate the time required to reach half the starting concentration. This equation is not just a mathematical tool but a reflection of how reaction mechanisms operate at the molecular level.
Steps to Derive the Second Order Reaction Half Life Equation
Understanding the second order reaction half life equation begins with deriving it from the fundamental principles of kinetics. Here are the key steps:
-
Start with the Rate Law: For a second-order reaction involving a single reactant A, the rate law is rate = -d[A]/dt = k[A]². The negative sign indicates that A is consumed over time.
-
Separate Variables
Rearrange the differential equation so that all terms containing [A] are on one side and the time variable is on the other:[ \frac{d[A]}{[A]^{2}} = -k,dt ]
This step isolates the concentration dependence, making the integration straightforward Worth keeping that in mind..
-
Integrate from t = 0 to t = t½
The limits of integration correspond to the initial concentration ([A]0) at time zero and the concentration at half‑life, ([A]{t_{1/2}} = \frac{[A]_0}{2}).[ \int_{[A]0}^{[A]0/2}\frac{d[A]}{[A]^{2}} = -k\int{0}^{t{1/2}} dt ]
Carrying out the integrals gives
[ \Bigl[-\frac{1}{[A]}\Bigr]_{[A]_0}^{[A]0/2}= -k,t{1/2} ]
[ \left(-\frac{2}{[A]_0}+ \frac{1}{[A]0}\right)= -k,t{1/2} ]
Simplifying:
[ \frac{1}{[A]0}= k,t{1/2} ]
-
Solve for the Half‑Life
The final expression for the half‑life of a second‑order reaction that depends on a single reactant is[ \boxed{t_{1/2}= \frac{1}{k,[A]_0}} ]
Notice that t½ is inversely proportional to the initial concentration. As ([A]_0) increases, the half‑life shortens, reflecting the concentration‑dependent nature of second‑order kinetics.
Extending the Derivation to Bimolecular Reactions (A + B → Products)
When two different reactants are involved, the rate law reads
[ \text{rate}= -\frac{d[A]}{dt}= k[A][B] ]
If the stoichiometry is 1:1 and the initial concentrations are ([A]_0) and ([B]_0), we can define a single variable (x) that represents the amount of A (and B) that has reacted at time t:
[ [A] = [A]_0 - x,\qquad [B] = [B]_0 - x ]
Substituting into the rate law and separating variables yields
[ \frac{dx}{([A]_0 - x)([B]_0 - x)} = k,dt ]
Integrating from 0 to (x_{1/2}) (the conversion that leaves half the limiting reactant) gives a more complex logarithmic expression:
[ t_{1/2}= \frac{1}{k([B]_0-[A]_0)}\ln!\left(\frac{[B]_0}{[A]_0}\right) ]
If the reactants start at equal concentrations ([A]_0=[B]_0), the equation collapses to the single‑reactant form shown above, because the denominator ([B]_0-[A]_0) becomes zero and the limit of the logarithmic term yields (1/([A]_0k)) Not complicated — just consistent..
Practical Implications of the Concentration‑Dependent Half‑Life
| Feature | First‑Order | Second‑Order (single reactant) | Second‑Order (A + B) |
|---|---|---|---|
| Half‑life expression | (t_{1/2}= \frac{\ln2}{k}) (constant) | (t_{1/2}= \frac{1}{k[A]_0}) (inversely proportional to ([A]_0)) | (t_{1/2}= \frac{1}{k([B]_0-[A]_0)}\ln!t is plotted |
| Dependence on initial concentration | None | Strong | Depends on the ratio of ([A]_0) to ([B]_0) |
| Typical mechanisms | Unimolecular decomposition, radioactive decay | Dimerization, recombination, second‑order hydrolysis | Bimolecular collisions, termolecular pathways reduced to effective bimolecular steps |
| Experimental observation | Straight line when (\ln[A]) vs. t is plotted | Straight line when (1/([B]-[A])) vs. |
Because the half‑life shortens as the reactant concentration rises, engineers must consider batch size and mixing efficiency when scaling up processes such as polymerizations, enzyme‑catalyzed syntheses, or pollutant degradation in water treatment. In a continuous‑flow reactor, the residence time may be set to a multiple of the calculated (t_{1/2}) to guarantee a desired conversion while avoiding excessive over‑reaction that could generate side products Most people skip this — try not to..
Common Pitfalls and How to Avoid Them
-
Assuming a Constant k
The rate constant k is temperature‑dependent (Arrhenius behavior) and may also be influenced by solvent polarity, ionic strength, or catalyst presence. Always verify that the experimental conditions match those under which k was determined. -
Neglecting the Stoichiometric Ratio in A + B Systems
If ([A]_0 \neq [B]_0), the limiting reactant dictates the half‑life. Using the single‑reactant formula in such cases can lead to under‑ or over‑estimation of reaction time No workaround needed.. -
Mixing Errors in Kinetic Experiments
In fast second‑order reactions, incomplete mixing can artificially inflate the apparent half‑life. Rapid quenching or stopped‑flow techniques help capture the true kinetic profile And that's really what it comes down to.. -
Units Mismatch
For second‑order reactions, k has units of M⁻¹·s⁻¹ (or L·mol⁻¹·s⁻¹). Plugging a first‑order k (s⁻¹) into the half‑life equation will produce nonsensical results It's one of those things that adds up..
Quick Checklist for Applying the Second‑Order Half‑Life Equation
- [ ] Verify the reaction follows second‑order kinetics (plot (1/[A]) vs. t; linearity confirms it).
- [ ] Determine whether the reaction is single‑reactant or bimolecular; choose the appropriate formula.
- [ ] Obtain a reliable k value at the experimental temperature (use Arrhenius if extrapolation is needed).
- [ ] Insert the correct initial concentration(s) and compute (t_{1/2}).
- [ ] Compare the calculated half‑life with experimental data; adjust for mixing or temperature deviations if necessary.
Real‑World Example: Hydrolysis of an Ester
Consider the base‑catalyzed hydrolysis of ethyl acetate:
[ \text{CH}_3\text{COOCH}_2\text{CH}_3 + \text{OH}^- ;\xrightarrow{k}; \text{CH}_3\text{COO}^- + \text{CH}_3\text{CH}_2\text{OH} ]
The reaction is second order overall (first order in ester, first order in hydroxide). Literature reports (k = 1.In real terms, 050\ \text{M}) and ([\text{OH}^-]_0 = 0. 050\ \text{M}). Suppose the experiment is conducted at 25 °C with ([ \text{EtOAc}]_0 = 0.2 \times 10^{2}\ \text{M}^{-1}\text{s}^{-1}) No workaround needed..
Because the initial concentrations are equal, the half‑life simplifies to
[ t_{1/2}= \frac{1}{k[\text{EtOAc}]_0}= \frac{1}{(1.2\times10^{2})(0.050)} \approx 0.17\ \text{s} ]
A rapid half‑life underscores why this reaction is often performed under controlled, low‑temperature conditions when selectivity is required. If the hydroxide concentration were halved while keeping the ester concentration constant, the half‑life would double, illustrating the direct influence of reactant concentration on reaction speed.
Summary and Outlook
The second‑order reaction half‑life equation encapsulates a fundamental truth of chemical kinetics: the rate at which a reactant disappears is intimately tied to how much of it is present. Practically speaking, unlike the comforting constancy of first‑order half‑lives, second‑order systems demand careful accounting of initial concentrations, stoichiometry, and temperature‑dependent rate constants. By mastering the derivation, recognizing the appropriate form for single‑reactant versus bimolecular reactions, and applying the checklist above, chemists and engineers can predict reaction times with confidence, design reactors that meet productivity targets, and troubleshoot kinetic anomalies when they arise It's one of those things that adds up..
In the broader context of modern science, this concentration‑dependent behavior is echoed in fields ranging from enzymology—where Michaelis–Menten kinetics reduce to second‑order forms at low substrate levels—to atmospheric chemistry, where pollutant removal often follows second‑order pathways. As computational tools and real‑time spectroscopic techniques continue to evolve, the classic half‑life equation remains a touchstone, reminding us that even the most sophisticated models ultimately rest on the simple, elegant mathematics first laid out by the pioneers of chemical kinetics That's the part that actually makes a difference. Took long enough..
Bottom line: For any reaction whose rate law is second order, the half‑life is not a fixed constant but a variable that shrinks as the reactant pool grows. Recognizing and applying this principle equips you to control, optimize, and scale chemical processes with precision It's one of those things that adds up..