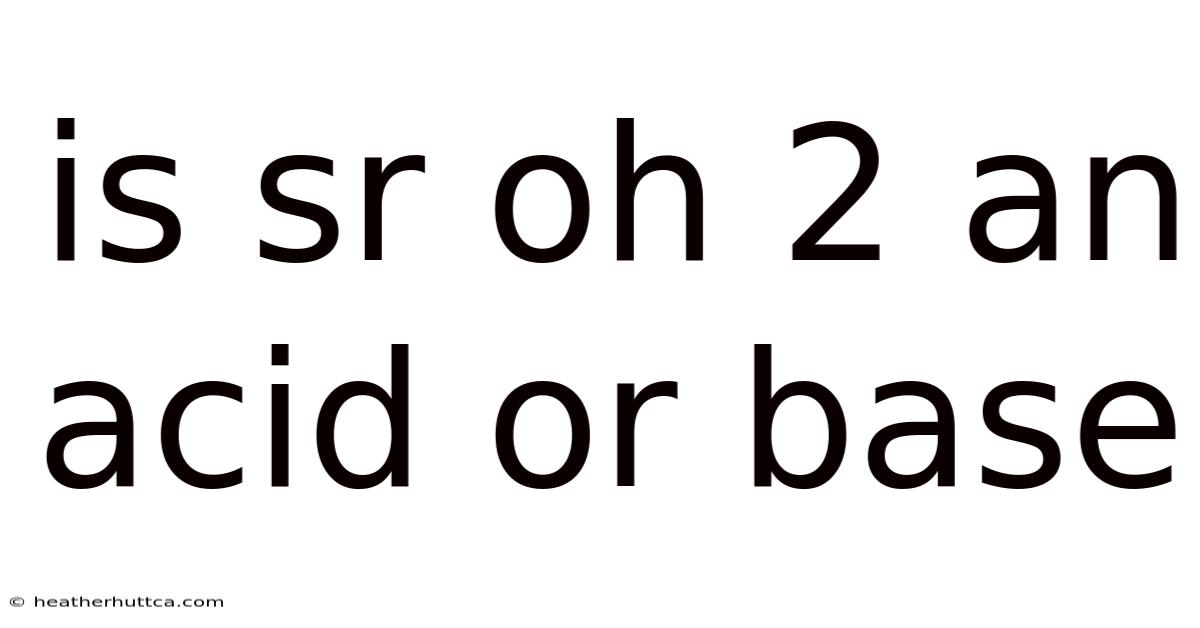Introduction
The question “Is Sr(OH)₂ an acid or a base?” may seem straightforward, but it opens a window into fundamental concepts of acid‑base chemistry, ionic dissociation, and the periodic trends that govern the behavior of metal hydroxides. Day to day, strontium hydroxide, Sr(OH)₂, is a white, crystalline solid that dissolves in water to give a strongly alkaline solution. Understanding why it behaves as a base—not an acid—requires a brief review of the Arrhenius, Brønsted–Lowry, and Lewis definitions of acids and bases, followed by an examination of the structure and solubility of Sr(OH)₂, its dissociation in aqueous media, and the resulting pH of its solutions.
In this article we will:
- Define acids and bases from multiple theoretical perspectives.
- Explore the periodic position of strontium and how it influences the properties of its hydroxide.
- Detail the dissolution and ionization of Sr(OH)₂ in water.
- Compare Sr(OH)₂ with other alkaline earth hydroxides.
- Address common misconceptions through a concise FAQ.
- Summarize the key take‑aways for students, teachers, and anyone curious about basic chemistry.
By the end of the reading, you will not only know that Sr(OH)₂ is a base, but also why it behaves that way and how its basicity can be quantified and applied in real‑world contexts.
1. Acid–Base Definitions: Setting the Framework
1.1 Arrhenius Concept
- Acid: Substance that increases the concentration of H⁺ ions in water.
- Base: Substance that increases the concentration of OH⁻ ions in water.
Under this classic model, Sr(OH)₂ clearly fits the definition of a base because it releases hydroxide ions when dissolved.
1.2 Brønsted–Lowry Concept
- Acid: Proton (H⁺) donor.
- Base: Proton acceptor.
When Sr(OH)₂ dissociates, the hydroxide ions (OH⁻) act as proton acceptors, capable of abstracting H⁺ from water or any acidic species, thereby confirming its basic character.
1.3 Lewis Concept
- Acid: Electron‑pair acceptor.
- Base: Electron‑pair donor.
The hydroxide ion possesses a lone pair of electrons that can be donated to a Lewis acid (e.g., Al³⁺, Fe³⁺). This electron‑pair donation further solidifies Sr(OH)₂’s status as a base in the Lewis framework And it works..
Bottom line: Regardless of which definition you adopt, Sr(OH)₂ behaves as a base.
2. Periodic Trends and the Nature of Strontium Hydroxide
2.1 Position of Strontium in the Periodic Table
Strontium (Sr) belongs to Group 2, the alkaline earth metals, situated directly below calcium (Ca) and above barium (Ba). Elements in this group share several characteristics:
- Low ionization energies relative to the main‑group metals.
- Tendency to form +2 cations (Sr²⁺).
- Formation of oxides (SrO) and hydroxides (Sr(OH)₂) that are basic rather than acidic.
2.2 Bonding in Sr(OH)₂
The compound consists of a Sr²⁺ cation electrostatically bound to two hydroxide anions (OH⁻). Because of that, 44). 95) and O (3.Which means the Sr–O bond is largely ionic because of the large difference in electronegativity between Sr (0. This ionic character facilitates easy dissociation in water, a prerequisite for basic behavior Surprisingly effective..
2.3 Solubility and Lattice Energy
Sr(OH)₂ is moderately soluble in water (≈ 0.5 g · 100 mL⁻¹ at 20 °C). But its lattice energy is lower than that of the corresponding calcium hydroxide (Ca(OH)₂) but higher than that of barium hydroxide (Ba(OH)₂). The balance between lattice energy and hydration energy determines its solubility, which in turn influences the concentration of OH⁻ released No workaround needed..
3. Dissolution and Ionization in Water
3.1 Overall Dissolution Reaction
[ \text{Sr(OH)}_2;(s) ;\xrightarrow{\text{H}_2\text{O}}; \text{Sr}^{2+};(aq) ;+; 2,\text{OH}^- ;(aq) ]
Each mole of solid yields two moles of hydroxide ions, doubling the alkalinity compared to a monobasic hydroxide Small thing, real impact..
3.2 Quantifying the Basicity
The K(_\text{sp}) (solubility product) for Sr(OH)₂ at 25 °C is approximately (3.2 \times 10^{-4}). Solving the equilibrium expression:
[ K_{\text{sp}} = [\text{Sr}^{2+}][\text{OH}^-]^2 ]
Let (s) be the molar solubility (mol L⁻¹). Then ([\text{Sr}^{2+}] = s) and ([\text{OH}^-] = 2s). Substituting:
[ 3.Consider this: 2 \times 10^{-4} = s (2s)^2 = 4s^3 \quad\Rightarrow\quad s = \sqrt[3]{\frac{3. 2 \times 10^{-4}}{4}} \approx 0.
Thus ([\text{OH}^-] = 2s \approx 0.086; \text{M}). The corresponding pOH is:
[ \text{pOH} = -\log_{10}(0.086) \approx 1.07 ]
And the pH (since (\text{pH} + \text{pOH} = 14) at 25 °C) is:
[ \text{pH} \approx 12.93 ]
A pH close to 13 confirms a strongly basic solution.
3.3 Comparison with Other Alkaline Earth Hydroxides
| Hydroxide | Solubility (g · 100 mL⁻¹, 20 °C) | Approx. 5 (very weakly basic) | | Ca(OH)₂ | 1.73 | ≈ 12.0009 | ≈ 10.Also, pH of Saturated Solution | |-----------|-----------------------------------|-----------------------------------| | Mg(OH)₂ | 0. 5 | ≈ 12.4 (moderately strong) | | Sr(OH)₂| 0.9 (strong) | | Ba(OH)₂ | 5.0 | ≈ 13.
The trend shows increasing basic strength down the group, reflecting decreasing lattice energy and increasing ionic size, which enable dissolution and OH⁻ release That alone is useful..
4. Practical Implications of Sr(OH)₂’s Basicity
4.1 Laboratory Uses
- pH Adjustment: Sr(OH)₂ can be employed when a high pH is required without introducing excessive metal ions that might interfere with certain analytical methods.
- Precipitation Reactions: Adding Sr(OH)₂ to solutions containing sulfate (SO₄²⁻) yields insoluble strontium sulfate (SrSO₄), a classic qualitative test for sulfate ions.
4.2 Industrial Applications
- Glass Manufacturing: Strontium compounds, including Sr(OH)₂, are used to modify the refractive index of glass, and the basic environment helps dissolve silica precursors.
- Ceramics and Pyrotechnics: The strong basicity aids in forming strontium‑based phosphors and colorants.
4.3 Safety Considerations
Because Sr(OH)₂ is a strong base:
- Corrosive: Direct contact can cause skin and eye irritation.
- Neutralization: Treat spills with dilute acetic acid or citric acid to convert OH⁻ to water and form harmless Sr²⁺ salts.
5. Frequently Asked Questions
Q1: Can Sr(OH)₂ act as an acid under any conditions?
A: No. Sr(OH)₂ does not donate protons; it only accepts them (as OH⁻). Even in highly acidic media, it will be protonated to form water and Sr²⁺, but it never behaves as a proton donor No workaround needed..
Q2: Is Sr(OH)₂ considered a “strong base” like NaOH?
A: In aqueous solution, Sr(OH)₂ is strongly basic because it dissociates completely to give OH⁻. Still, its solubility is lower than NaOH, so the maximum achievable OH⁻ concentration is less than that of a saturated NaOH solution.
Q3: Why does Sr(OH)₂ have a lower solubility than Ba(OH)₂ despite being lower in the group?
A: Solubility depends on a balance between lattice energy and hydration energy. While Ba²⁺ is larger (lower lattice energy), its hydration energy is also lower, leading to a net increase in solubility compared to Sr²⁺ Nothing fancy..
Q4: Can Sr(OH)₂ be used to prepare strontium carbonate (SrCO₃)?
A: Yes. Bubbling CO₂ through an aqueous Sr(OH)₂ solution precipitates SrCO₃:
[ \text{Sr(OH)}_2 + \text{CO}_2 \rightarrow \text{SrCO}_3 \downarrow + \text{H}_2\text{O} ]
Q5: What is the effect of temperature on the basicity of Sr(OH)₂ solutions?
A: Solubility of Sr(OH)₂ increases with temperature, allowing more OH⁻ to be released, which slightly raises the pH of saturated solutions. That said, the pH remains near 13 across typical laboratory temperature ranges.
6. Conclusion
Strontium hydroxide, Sr(OH)₂, unequivocally behaves as a base across all major acid‑base theories. Its ionic composition, the ease with which it dissociates into Sr²⁺ and two hydroxide ions, and the resulting high pH of its aqueous solutions all point to strong basicity. Periodic trends explain why its basic strength sits between calcium hydroxide and barium hydroxide, while its moderate solubility makes it a useful reagent in both laboratory and industrial settings.
Understanding the nature of Sr(OH)₂ deepens appreciation for how the periodic table, lattice energetics, and solvation dynamics intertwine to dictate whether a compound is acidic, neutral, or basic. Whether you are a student preparing for an exam, a teacher designing a lab, or a professional seeking a reliable alkaline reagent, recognizing Sr(OH)₂ as a strong base equips you with the knowledge to handle it safely and exploit its properties effectively Worth knowing..