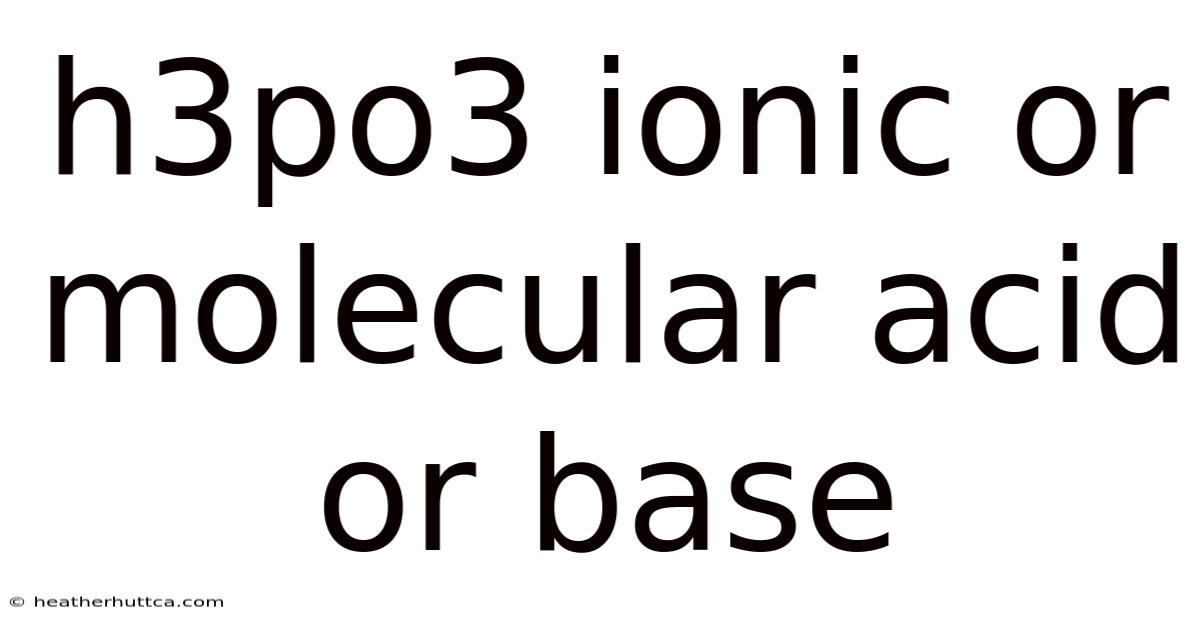Understanding H₃PO₃: Is It an Ionic or Molecular Acid?
Phosphorous acid, H₃PO₃, often sparks confusion among chemistry students and professionals alike: is it an ionic compound, a molecular acid, or even a base? This full breakdown clears the fog by exploring the structure, bonding, and behavior of H₃PO₃ in water and other media. By the end, you’ll know exactly how to classify phosphorous acid, why it behaves the way it does, and how its properties compare to other common acids and bases That alone is useful..
Introduction: Why the Classification Matters
Correctly labeling a substance as ionic, molecular, acidic, or basic is more than academic semantics. It determines:
- Solubility predictions – ionic compounds dissolve readily in polar solvents, while molecular acids may show limited solubility.
- Reactivity patterns – acids donate protons, bases accept them; ionic species often undergo double‑replacement reactions.
- Safety handling – the strength of an acid influences protective measures in the laboratory.
Phosphorous acid appears in textbooks as a “diprotic acid” (it can donate two protons) but its formula, H₃PO₃, suggests three hydrogen atoms. Understanding the underlying bonding and ionization is essential for accurate classification Worth keeping that in mind..
1. Molecular Structure of H₃PO₃
1.1. Lewis Diagram and Geometry
Phosphorous acid is best represented by the structural formula HO–P(=O)(OH)–H. Key points:
- Central atom: phosphorus (P) in the +3 oxidation state.
- Bonding: two hydroxyl groups (–OH) attached to phosphorus, one double‑bonded oxygen (=O), and one hydrogen directly bonded to phosphorus (P–H).
- Molecular geometry: a trigonal pyramidal arrangement around phosphorus, similar to that of ammonia (NH₃), due to the lone pair on P.
The presence of covalent P–O and P–H bonds confirms that H₃PO₃ is fundamentally a covalent (molecular) compound rather than an ionic lattice.
1.2. Why It Is Not a Simple Ionic Salt
If H₃PO₃ were ionic, we would expect a formula like H⁺ + PO₃³⁻ (phosphite ion). However:
- The phosphite ion (PO₃³⁻) exists only after deprotonation of phosphorous acid.
- In the neutral molecule, the phosphorus–hydrogen bond is covalent, and the hydroxyl groups are not fully ionized.
- Crystallographic data show discrete molecules held together by hydrogen bonds, not a crystal lattice of separate cations and anions.
Thus, the parent compound is molecular, and its ionic nature emerges only after dissociation in water.
2. Acidic Behavior: Proton Donation in Water
2.1. First and Second Dissociation Steps
When H₃PO₃ dissolves in water, two of its three hydrogens can dissociate as protons (H⁺). The stepwise ionization is:
-
First dissociation (stronger):
[ \text{H₃PO₃} ;\rightleftharpoons; \text{H⁺} + \text{H₂PO₃⁻} ] pK₁ ≈ 1.3 – a relatively strong acid for a non‑oxide acid. -
Second dissociation (weaker):
[ \text{H₂PO₃⁻} ;\rightleftharpoons; \text{H⁺} + \text{HPO₃^{2-}} ] pK₂ ≈ 6.7 – comparable to the acidity of weak organic acids like acetic acid.
The third hydrogen (the one directly bound to phosphorus) does not ionize under normal aqueous conditions; the P–H bond remains covalent, making H₃PO₃ a diprotic acid, not triprotic.
2.2. Comparison with Phosphoric Acid (H₃PO₄)
- Phosphoric acid (H₃PO₄) is a triprotic acid with three ionizable protons (pK₁≈2.1, pK₂≈7.2, pK₃≈12.3).
- Phosphorous acid lacks the third ionizable hydrogen because the P–H bond is much less acidic than P–OH bonds.
Understanding this distinction reinforces that H₃PO₃’s acidity stems from molecular dissociation, not from an inherent ionic lattice.
3. Ionic Species Formed After Dissociation
Once dissolved, H₃PO₃ yields ionic species:
- Hydrogen phosphite ion (H₂PO₃⁻)
- Phosphite ion (HPO₃²⁻) after the second deprotonation
These ions are anion counterparts of the original molecular acid. Their existence in solution demonstrates that both molecular and ionic forms coexist depending on the degree of proton loss.
4. Is H₃PO₃ a Base? – Evaluating Basicity
A base is defined by its ability to accept protons (Bronsted–Lowry) or donate electron pairs (Lewis). Phosphorous acid:
- Does not accept protons under normal conditions; instead, it donates them.
- Lacks a lone pair on a nitrogen or oxygen atom that could act as a strong Lewis base.
As a result, H₃PO₃ is not a base. Its conjugate base, the phosphite ion (HPO₃²⁻), can act as a weak base, but the parent acid itself is unequivocally acidic And that's really what it comes down to. That alone is useful..
5. Practical Implications of Its Classification
5.1. Laboratory Handling
- Acidic nature: Use gloves, goggles, and a fume hood when handling concentrated solutions.
- Molecular character: Expect moderate volatility; H₃PO₃ is a colorless liquid with a faint odor, not a solid salt.
5.2. Industrial Uses
- Reducing agent: The P–H bond can donate hydrogen in redox reactions, valuable in metal plating and organic synthesis.
- Buffer component: Because of its two dissociation constants, phosphorous acid can help maintain pH in the range of 2–8 when combined with appropriate salts.
5.3. Environmental Considerations
- Phosphite ions produced after dissociation are less toxic than orthophosphate (PO₄³⁻) but can still contribute to phosphorus cycling in ecosystems.
6. Frequently Asked Questions (FAQ)
Q1: Can H₃PO₃ be considered an ionic compound in the solid state?
A: No. In the solid state, phosphorous acid forms a hydrogen‑bonded molecular crystal, not an ionic lattice. Only after dissolution do ionic species appear Nothing fancy..
Q2: Why does H₃PO₃ have only two ionizable hydrogens?
A: The hydrogen attached directly to phosphorus (P–H) is covalent and has a very high bond dissociation energy, making it non‑acidic under normal conditions Worth keeping that in mind..
Q3: Is phosphorous acid stronger or weaker than phosphoric acid?
A: The first dissociation of H₃PO₃ (pK₁≈1.3) is stronger than the first dissociation of H₃PO₄ (pK₁≈2.1). Even so, phosphoric acid can donate three protons, whereas phosphorous acid can donate only two And it works..
Q4: What happens if H₃PO₃ reacts with a strong base like NaOH?
A: It undergoes neutralization, forming sodium phosphite salts:
[
\text{H₃PO₃} + 2\text{NaOH} \rightarrow \text{Na₂HPO₃} + 2\text{H₂O}
]
The product is an ionic salt (sodium phosphite).
Q5: Does H₃PO₃ exhibit any amphoteric behavior?
A: Not as a neutral molecule. Its conjugate base (phosphite ion) can act as a weak base, but the acid itself does not display amphoterism.
7. Summary: The Bottom Line
- Molecular nature: H₃PO₃ is a covalent molecule with a trigonal pyramidal geometry. It is not an ionic solid in its pure form.
- Acidic character: It behaves as a diprotic Brønsted–Lowry acid, donating two protons in aqueous solution and forming hydrogen phosphite (H₂PO₃⁻) and phosphite (HPO₃²⁻) ions.
- Not a base: The molecule does not accept protons; only its conjugate bases can act as weak bases.
- Practical relevance: Its reducing ability, buffering capacity, and moderate acidity make it valuable in industrial and laboratory contexts.
Understanding that phosphorous acid is a molecular diprotic acid—with ionic behavior emerging only after dissolution—provides a solid foundation for both academic study and real‑world applications. The distinction between its molecular structure and the ions it generates is a classic illustration of how chemical classification depends on the environment (solid vs. solution) and underscores the importance of precise terminology in chemistry Most people skip this — try not to..
8. Further Exploration
Beyond its fundamental properties, phosphorous acid and its derivatives find application in a diverse range of fields. In the pharmaceutical industry, phosphite salts are utilized as stabilizers and preservatives, particularly in injectable medications. Their ability to scavenge free radicals makes them valuable in antioxidant formulations. In agriculture, phosphite compounds are incorporated into fertilizers to enhance phosphorus availability to plants. Adding to this, they play a crucial role in the production of flame retardants, owing to their ability to interfere with combustion processes. Practically speaking, the research into novel phosphite-based materials is ongoing, with scientists exploring their potential in areas like battery technology and advanced polymers. The nuanced chemical behavior of phosphorous acid, bridging the gap between covalent bonding and ionic dissociation, continues to be a subject of active investigation, promising further breakthroughs in various scientific disciplines.
9. Conclusion
All in all, phosphorous acid (H₃PO₃) presents a fascinating case study in chemical behavior. Its molecular structure, characterized by covalent bonds and a trigonal pyramidal geometry, dictates its properties as a diprotic Brønsted-Lowry acid. While not an ionic compound in the solid state, its dissociation in aqueous solution yields a spectrum of ionic species, each with distinct chemical characteristics and applications. Understanding the distinction between the molecule itself and the ions it forms is essential for accurate chemical interpretation and informed application. Which means the study of phosphorous acid highlights the importance of considering the environment – whether solid or solution – when classifying chemical entities and appreciating the complex relationship between molecular structure and chemical reactivity. Its unique properties make it a valuable compound with ongoing relevance across scientific and industrial landscapes.